Research
| In the Yaghi group we are building chemical structures by stitching molecules together into large and extended frameworks within which we can store hydrogen, methane, and separate carbon dioxide. The interior of the crystals is capable of compacting gases under ambient conditions thus foregoing the use of high pressures and low temperatures. In this movie, crystals of metal-organic framework-5 (MOF-5, numbered in roughly chronological order of discovery) are made and their structure is precisely designed to have zinc oxide units linked by organic struts (terephthalate) to make a porous network into which voluminous gases can be compacted and transported. This large open space is currently being used for positioning of organic and organometallic catalysts, charge storage for supercapacitors and binding of biological molecules such as proteins and metabolites. The movie was made by our industry partner BASF (Germany). |
Reticular Chemistry
 |
| The six structures placed in the center are materials invented by the Yaghi group. From left to right in the top row are metal-organic frameworks, covalent organic frameworks, and zeolitic imidazolate frameworks, and in the bottom row metal triazolates, metal-organic polyhedra, and metal catecholates. The panel on the left is a figure of a multivariable metal-organic framework found to have sequences of functionalities capable of highly selective separation of carbon dioxide, while the panel on the right is a covalent organic framework composed entirely of light elements (B, O, C) and represents a new class of porous crystals. In the Yaghi group students are free to explore new directions in reticular materials research and experience the thrill of discovery by making new forms of matters and studying their benefit to society. |
The Yaghi group is developing the science and applications of precise assembly of materials by the molecular building block approach. To do this, we use reticular chemistry where molecular building blocks (organic molecules, inorganic clusters and complexes, proteins, peptides, and dendrimers) are linked into extended frameworks using strong bonds. This chemistry allows us to translate the high functionality of molecules into solids without losing the robustness needed for making useful materials, or the dynamics and molecular flexibility required for highly functional materials. Thus in our group new materials are created by 'stitching' metal-ions and metal complexes with organic linkers to make extended porous frameworks called metal-organic frameworks (MOFs), and zeolitic imidazolate frameworks (ZIFs). We also link organic units together to make covalent organic frameworks (COFs). This approach is elaborated to make metal-catecholates (CATs) and metal triazolates (METs). These are all new classes of porous crystals made by our group, and studied for their applications to clean energy storage and generation, clean water generation and delivery, supercapacitors, thermal batteries, ion-conductivity, electronic conductivity, and drug delivery.
- Metal-organic frameworks: assembly of transition metal ions and main group ions with organic linkers (Read more: Nature, YouTube video, C&EN)
- Covalent organic frameworks: assembly of organic building units held together by strong covalent bonds and light elements (B, O, C, N) (Read more: C&EN, Science)
- Zeolitic imidazolate frameworks: assembly of imidazolate (and its derivatives) and transition metal ions as well as main group elements (Read more: RSC website)
- Metal-organic polyhedra: assembly of metal ions and organic links to make discrete porous molecules with faceted polyhedra
- New porous crystals based on catecholates, triazine and other link functionalities including peptides and proteins to make porous bioframeworks
- Developing strategies for making crystalline solids with ultra-high porosity and very low density, and their use as gas storage materials and as electronic materials (Read more: C&EN #1, C&EN #2)
- The Chemistry and Applications of Metal-Organic Frameworks, H. Furukawa, K. E. Cordova, M. O'Keeffe, O. M. Yaghi, Science, 2013, 341, 1230444.

- New Porous Crystals of Extended Metal-Catecholates, M. Hmadeh, Z. Lu, Z. Liu, F. Gándara, H. Furukawa, S. Wan, V. Augustyn, R. Chang, L. Liao, F. Zhou, E. Perre, V. Ozolins, K. Suenaga, X. Duan, B. Dunn, Y. Yamamto, O. Terasaki, O. M. Yaghi, Chem. Mater., 2012, 24, 3511-3513.

- Porous, Conductive Metal-Triazolates and Their Structural Elucidation by the Charge-Flipping Method, F. Gándara, F. J. Uribe-Romo, D. K. Britt, H. Furukawa, L. Lei, R. Cheng, X. Duan, M. O'Keeffe, O. M. Yaghi, Chem. Eur. J., 2012, 18, 10595-10601.

- Ultra-High Porosity in Metal-Organic Frameworks, H. Furukawa, N. Ko, Y. B. Go, N. Aratani, S. B. Choi, E. Choi, A. O. Yazaydin, R. Q. Snurr, M. O'Keeffe, J. Kim, O. M. Yaghi, Science, 2010, 239, 424-428.

- Synthesis, Structure, and Carbon Dioxide Capture Properties of Zeolitic Imidazolate Frameworks, A. Phan, C. J. Doonan, F. J. Uribe-Romo, C. B. Knobler, M. O'Keeffe, O. M. Yaghi, Acc. Chem. Res., 2010, 43, 58-67.

- Reticular Chemistry of Metal-Organic Polyhedra, D. J. Tranchemontagne, Z. Ni, M. O'Keeffe, O. M. Yaghi, Angew. Chem. Int. Ed., 2008, 47, 5136-5147.

- Designed Synthesis of 3D Covalent Organic Frameworks, H. M. El-Kaderi, J. R. Hunt, J. L. Mendoza-Cortés, A. P. Côté, R. E. Taylor, M. O'Keeffe, O. M. Yaghi, Science, 2007, 316, 268-272.

- Reticular Synthesis and the Design of New Materials, O. M. Yaghi, M. O'Keeffe, N. W. Ockwig, H. K. Chae, M. Eddaoudi, J. Kim, Nature, 2003, 423, 705-714.

Water harvesting from desert air
 |
| A hand-held device filled with a porous metal-organic framework, MOF-303, is capable of harvesting water from desert air without any power or energy input aside from the ambient sunlight at Death Valley, a location known for being the hottest place on Earth and the driest desert in North America. This photo shows a prototype of the device designed by Yaghi Group positioned at Furnace Creek— within Death Valley — in California. |
The majority of the world population lives in water-stressed regions where access to fresh and clean
water is limited. Nevertheless, our atmosphere contains about 13,000 trillion liters of water vapor at any
given time, more than all rivers and lakes combined. Although many solutions have been proposed for
harvesting water from fog and high humidity climates, to date no efficient technology exists for harvesting
water from arid regions of the world where it is most needed. In 2014, the Yaghi Group showed in a
groundbreaking report that MOFs and COFs can be tailored to harvest drinking water from desert air.
Further physical characterization revealed that the high water affinity of these MOF materials is due to
water binding to their pores in exactly the same way as water molecules in ice. In essence, inside these
MOFs, one has solid water (i.e. ice fragments) forming at room temperature. To get the water out, as the
goal is liquid water, one simply heats the materials to a mere 45 ºC. This provides enough energy to
release the water from the pores to make clean water. The power of MOFs and COFs for harvesting
water from air was demonstrated when Yaghi's group collected drinkable amount of water with MOF
water harvesters in both Arizona and Death Valley deserts during the hottest and driest time of the year
with no external power source. Our MOF water harvesting technology has been showcased at the 2017
World Economic Forum as one of the top ten emerging technologies to change the world. IUPAC in 2019
also named our MOF water harvesting one of the top 10 breakthroughs in chemistry.
- Design and synthesis of MOFs and COFs for water harvesting (Read more: Innovation & Tech Today)
- Physical characterization of the mechanisms of water sorption in MOFs and COFs (Read more: Berliner Zeitung)
- MOF/COF water harvester device engineering (Read more: Science)
- Field trials of harvesting water from desert air (Read more: Nature, Berkeley News)
- MOF water harvester produces water from Death Valley desert air in ambient sunlight, W. Song, Z. Zheng, A. H. Alawadhi, O. M. Yaghi, Nat. Water, 2023, 1, 626-634.

- Evolution of water structures in metal-organic frameworks for improved atmospheric water harvesting, N. Hanikel, X. Pei, S. Chheda, H. Lyu, W. Jeong, J. Sauer, L. Gagliardi, O. M. Yaghi, Science, 2021, 374, 454-459.

- Rapid Cycling and Exceptional Yield in a Metal-Organic Framework Water Harvester, N. Hanikel, M. S. Prévot, F. Fathieh, E. A. Kapustin, H. Lyu, H. Wang, N. J. Diercks, T. G. Glover, O. M. Yaghi, ACS Cent. Sci., 2019, 5, 1699-1706.

- Practical Water Production from Desert Air, F. Fathieh, M. J. Kalmutzki, E. A. Kapustin, P. J. Waller, J. Yang, O. M. Yaghi, Sci. Adv., 2018, 4, eaat3198.

- Water Harvesting from Air with Metal-Organic Frameworks Powered by Natural Sunlight, H. Kim, S. Yang, S. R. Rao, S. Narayanan, E. A. Kapustin, H. Furukawa, A. S. Umans, O. M. Yaghi, E. N. Wang, Science, 2017, 356, 430-434.

- Water Adsorption in Porous Metal-Organic Frameworks and Related Materials, H. Furukawa, F. Gándara, Y.-B. Zhang, J. Jiang, W. L. Queen, M. R. Hudson, O. M. Yaghi, J. Am. Chem. Soc., 2014, 136, 4369-4381.

Carbon dioxide capture
 |
| Capturing carbon from the air just got easier. A new type of porous material called a covalent organic framework quickly sucks up carbon dioxide from ambient air. Pictured is a vial of COF- 999, which is yellow, with UC Berkeley’s landmark campanile in the background. |
Capturing and storing the carbon dioxide humans produce is key to lowering atmospheric greenhouse
gases and slowing global warming. Today’s carbon capture technologies work well only for concentrated
sources of carbon, such as power plant exhaust. The same methods cannot efficiently capture carbon
dioxide from ambient air, where concentrations are hundreds of times lower than in flue gases. Yet direct
air capture, or DAC, is being counted on to reverse the rise of CO2 levels, which have reached 426 parts
per million (ppm), 50% higher than levels before the Industrial Revolution. Without it, according to the
Intergovernmental Panel on Climate Change, we won’t reach humanity’s goal of limiting warming to 1.5
°C (2.7 °F) above preexisting global averages. New type of carbon dioxide absorbing porous materials,
MOFs and COFs, are being developed by the Yaghi Lab. These porous materials capture CO2 from point
sources or ambient air without degradation by water or other contaminants, one of the limitations of
existing carbon capture technologies.
- Design and synthesis of MOFs and COFs for CO2 capture from flue gas (Read more: Berkeley Lab, ABC News, The New York Times, The New York Times, Chemistry World, Youtube, Science News, Science)
- Design and synthesis of MOFs and COFs for direct open-air capture of carbon dioxide (Read more: SFGATE, CBS News, Los Angeles Times)
- Physical characterization of the mechanisms of carbon capture in MOFs and COFs
- Catalytic CO2 Reduction in MOFs and COFs (Read more: MIT Technology Review, X)
- MOF/COF carbon capture device engineering
- Carbon dioxide capture from open air using covalent organic frameworks, Z. Zhou, T. Ma, H. Zhang, S. Chheda, H. Li, K. Wang, S. Ehrling, R. Giovine, C. Li, A. H. Alawadhi, M. M. Abduljawad, M. O. Alawad, L. Gagliardi, J. Sauer, O. M. Yaghi, Nature, 2024, 635, 96–101.

- Covalent Organic Frameworks for Carbon Dioxide Capture from Air, H. Lyu, H. Li, N. Hanikel, K. Wang, O. M. Yaghi, J. Am. Chem. Soc., 2022, 144, 12989–12995.

- Carbon Dioxide Capture Chemistry of Amino Acid Functionalized Metal–Organic Frameworks in Humid Flue Gas, H. Lyu, O. I. Chen, N. Hanikel, M. I. Hossain, R. W. Flaig, X. Pei, A. Amin, M. D. Doherty, R. K. Impastato, T. G. Glover, D. R. Moore, O. M. Yaghi, J. Am. Chem. Soc., 2022, 144, 2387–2396.

- Covalent Organic Frameworks Comprising Cobalt Porphyrins for Catalytic CO2 Reduction in Water, S. Lin, C. S. Diercks, Y.-B. Zhang, N. Kornienko, E. M. Nichols, Y. Zhao, A. R. Paris, D. Kim, P. Yang, O. M. Yaghi, C. J. Chang, Science, 2015, 349, 1208-1213.

- Metal-Organic Frameworks with Precisely Designed Interior for Carbon Dioxide Capture in the Presence of Water, A. M. Fracaroli, H. Furukawa, M. Suzuki, M. Dodd, S. Okajima, F. Gándara, J. A. Reimer, O. M. Yaghi, J. Am. Chem. Soc., 2014, 136, 8863-8866.

- Highly Efficient Separation of Carbon Dioxide by a Metal-Organic Framework Replete with Open Metal Sites, D. Britt, H. Furukawa, B. Wang, T. G. Glover, O. M. Yaghi, Proc. Natl. Acad. Sci. U.S.A., 2009, 106, 20637-20640.

- Colossal Cages in Zeolitic Imidazolate Frameworks as Selective Carbon Dioxide Reservoirs, B. Wang, A. P. Côté, H. Furukawa, M. O'Keeffe, O. M. Yaghi, Nature, 2008, 453, 207-211.

- High-Throughput Synthesis of Zeolitic Imidazolate Frameworks and Application to CO2 Capture, R. Banerjee, A. Phan, B. Wang, C. Knobler, H. Furukawa, M. O'Keeffe, O. M. Yaghi, Science, 2008, 319, 939-943.

- Metal-Organic Frameworks with Exceptionally High Capacity for Storage of Carbon Dioxide at Room Temperature, A. R. Millward, O. M. Yaghi, J. Am. Chem. Soc., 2005, 127, 17998-17999.

Molecular weaving
 |
| COF-505 is the first materials to be woven at the atomic and molecular Levels. It is a 3D covalent organic framework made by weaving together molecular helical organic threads, a fabrication technique that yields significant advantages in structural flexibility, resiliency and reversibility over previous COFs. |
There are many different ways to make materials on the nano or molecular scale but weaving, the oldest
and most enduring method of making fabrics, has not been one of them - until the Yaghi Group in 2016
created the first materials woven at the atomic and molecular Levels, COF-505. In this first study, the
Yaghi Group used a copper(I) complex as a template for bringing threads of the organic compound
“phenanthroline” into a woven pattern to produce an immine-based framework they dubbed COF-505.
Through X-ray and electron diffraction characterizations, the researchers discovered that the copper(I)
ions can be reversibly removed or restored to COF-505 without changing its woven structure.
Demetalation of the COF resulted in a tenfold increase in its elasticity and remetalation restored the COF
to its original stiffness. We took advantage of the unusual property of molecular weaving to make polymer
composites stronger, tougher, and more resistant to fracture by threading polymer strands through the
woven network. When we add a small amount (1%) of these woven COF crystals to other materials such
as polymer or plastic in this case, the materials become significantly tougher and can have a high
tolerance for damages and fractures. This could have a huge impact on the materials industry. Using
reticular synthesis, we have also created a new type of 2D and 3D materials from millions of identical,
interlocking molecules that are flexible, strong and resilient, like the chain mail that protected medieval
knights.
- Strategies of molecular weaving in MOFs and COFs (Read more: Science)
- Molecular weaving as materials (Read more: Berkeley Chemistry News)
- New flexible and resilient structures (Read more: C&EN)
- The propensity for covalent organic frameworks to template polymer entanglement, S. E. Neumann, J. Kwon, C. Gropp, L. Ma, R. Giovine, T. Ma, N. Hanikel, K. Wang, T. Chen, S. Jagani, R. O. Ritchie, T. Xu, O. M. Yaghi, Science, 2024, 383, 626-634.

- Molecular weaving of chicken-wire covalent organic frameworks, X. Han, T. Ma, B. L. Nannenga, X. Yao, S. E. Neumann, P. Kumar, J. Kwon, Z. Rong, K. Wang, Y. Zhang, J. A. R. Navarro, R. O. Ritchie, Y. Cui, O. M. Yaghi, Chem, 2023, 9, 2509-2517.

- Catenated covalent organic frameworks constructed from polyhedra, T. Ma, Y. Zhou, C. S. Diercks, J. Kwon, F. Gándara, H. Lyu, N. Hanikel, P. Pena-Sánchez, Y. Liu, N. J. Diercks, R. O. Ritchie, D. M. Proserpio, O. Terasaki, O. M. Yaghi, Nat. Synth., 2023, 2, 286-295.

- Molecular Weaving of Covalent Organic Frameworks for Adaptive Guest Inclusion, Y. Liu, Y. Ma, J. Yang, C. S. Diercks, N. Tamura, F. Jin, O. M. Yaghi, J. Am. Chem. Soc., 2018, 140, 16015-16019.

- Weaving of Organic Threads into a Crystalline Covalent Organic Framework, Y. Liu, Y. Ma, Y. Zhao, X. Sun, F. Gándara, H. Furukawa, Z. Liu, H. Zhu, C. Zhu, K. Suenaga, P. Oleynikov, A. S. Alshammari, X. Zhang, O. Terasaki, O. M. Yaghi, Science, 2016, 351, 365-369.

AI & Digital discovery cycle
 |
| We have trained Large language models (LLMs) such as ChatGPT to perform the research tasks of text mining, image mining, advising, supervising, molecular editing, essentially functioning as a research group. |
We develop generative artificial intelligence (AI) models and use large language models (LLMs) to
accelerate discovery of MOFs and COFs in reticular chemistry. These models are used to augment our
laboratory research, helping us build knowledge from literature and our own laboratory for design of
materials, and to collect and interpret experimental data. We are working intensively on developing these
concepts and methods for their routine use in scientific research, including automated prompt
engineering, knowledge and tool augmentation, and for fine-tuning reticular materials for applications to
water harvesting and carbon capture. Our recent results show how these 'chemistry-aware' models can
be integrated into existing practices of reticular chemistry and tailored to specific tasks, transforming the
traditional 'make, characterize, use' protocol driven by empirical knowledge into a discovery routine based
on finding synthesis-structure-property-performance relationships. These modularized LLM agents with
tool making and using ability can be subsequently integrated in a multi-agentic system within a laboratory
environment to collaborate with chemists to streamline labor-intensive research tasks and accelerate the
materials discovery cycle. We are exploring how LLMs can lower the barriers to applying generative AI
and data-driven workflows to address the crystallization challenges of reticular structures. On a
fundamental level, we wish to equip chemists, both computational and experimental, with the insights
necessary to harness the capabilities of AI for advancing material discovery and enable what we call the
'digital discovery cycle'. Here, robotics, image recognition, building datasets, and using machine learning
are combined to discover new materials for tackling the climate problem and to do so better and faster
than what we can do today.
- Generative artificial intelligence (AI) for MOF and COF discoveries
- Large language models (LLMs): Text Mining, Image Mining, ChatGPT Supervisor, Molecular Editing, ChatGPT Research Group (Read more: Phys.org)
- Robotics
- Machine Learning
- Image and data mining in reticular chemistry powered by GPT-4V, Z. Zheng, Z. He, O. Khattab, N. Rampal, M. A. Zaharia, C. Borgs, J. T. Chayes, O. M. Yaghi, Digital Discovery, 2024, 3, 491-501.

- Shaping the Water-Harvesting Behavior of Metal-Organic Frameworks Aided by Fine-Tuned GPT Models, Z. Zheng, A. H. Alawadhi, S. Chheda, S. E. Neumann, N. Rampal, S. Liu, H. L. Nguyen, Y. Lin, Z. Rong, J. I. Siepmann, L. Gagliardi, A. Anandkumar, C. Borgs, J. T. Chayes, O. M. Yaghi, J. Am. Chem. Soc., 2023, 145, 28284-28295.

- ChatGPT Research Group for Optimizing the Crystallinity of MOFs and COFs, Z. Zheng, O. Zhang, H. L. Nguyen, N. Rampal, A. H. Alawadhi, Z. Rong, T. Head-Gordon, C. Borgs, J. T. Chayes, O. M. Yaghi, ACS Cent. Sci., 2023, 9, 2161-2170.

- A GPT-4 Reticular Chemist for Guiding MOF Discovery, Z. Zheng, Z. Rong, N. Rampal, C. Borgs, J. T. Chayes, O. M. Yaghi,, Angew. Chem. Int. Ed., 2023, 62, e2023119.

- ChatGPT chemistry assistant for text mining and the prediction of MOF synthesis, Z. Zheng, O. Zhang, C. Borgs, J. T. Chayes, O. M. Yaghi, J. Am. Chem. Soc., 2023, 145, 18048-18062.

CLEAN ENERGY
 |
| From left to right are illustrations of gases/molecules such as hydrogen, methane, carbon dioxide, and water which the Yaghi group has been studying and has uncovered their selective uptake into metal-organic frameworks (MOFs) and other reticular materials. Highly crystalline and well-shaped crystals can be made in our laboratory. Through our long-standing collaboration with BASF, it has become possible to make these materials in ton quantities. We have shown that fuel tanks filled with our materials can store double, and in many cases, triple the amount of methane and such automobiles were driven 28,000 km in four continents by German explorers without loss of the efficiency of MOF fuel tanks thus demonstrating that MOF fuel tanks are competitive with CNG. On the right are shown crystals of a MOF where our group has been able to expose metal sites and for the first time pin-point their location by single crystal x-ray diffraction techniques and demonstrate their efficiency in capturing carbon dioxide. In the Yaghi group students are working on materials beyond MOFs (e.g. COFs, ZIFs and METs) for clean energy applications. |
The Yaghi group was the first to uncover the porosity of MOFs. In 1998 and 1999, we reported their synthesis and architectural stability. Using gas adsorption isotherms we showed that zinc terephthalate MOFs (MOF-2 and MOF-5) retain their open structure in the absence of guests and that gases can pass in and out of the pores with full retention of the MOF structure. Since then we showed that MOFs can be functionalized and their components (metal containing units and organic links) can be widely varied. This made available a large class of MOF materials whose gas adsorption properties continue to be studied by our group. Specifically, we have several ongoing programs focused on hydrogen storage and methane storage and transport for automobile fueling. We also work extensively on carbon capture from the atmosphere, automobiles and power plants. More recently, we have initiated research programs where it is possible to use amyloids (long known to be important in the onslaught of diseases such as Alzheimer's) for demonstrating that lysine functionalities can trap carbon dioxide in the presence of water. MOFs are also being investigated in our group for their applications to clean water using artificial leaf constructs, and as thermal batteries for heating and cooling automobiles.
- Exceptional hydrogen uptake capacity in MOFs, COFs, ZIFs, and their functionalized forms. New strategies for room temperature storage of hydrogen in metal cluster- containing MOFs (Read more: C&EN, RSC website)
- MOFs, COFs, and ZIFs for methane storage and separation from natural gas. Use of methane filled MOFs for automobile fueling (Read more: C&EN)
- Carbon dioxide capture from the atmosphere, automobiles and power plants using MOFs, COFs, and ZIFs. Developing strategies derived from —biological systems where carbon dioxide can be stored and fixed to make fuels (Read more: the New York Times #1, the New York Times #2, RSC website #1, RSC website #2)
- Use of MOFs to trap water from the atmosphere and releasing it as means of purification and transport (i.e. dehumidification for drinking water without electric power)
- Applications of MOFs with ultra-high capacity for water in thermal battery applications for heat exchange in automobiles (Read more: SAE International)
- Metal-Organic Frameworks with Precisely Designed Interior for Carbon Dioxide Capture in the Presence of Water, A. M. Fracaroli, H. Furukawa, M. Suzuki, M. Dodd, S. Okajima, F. Gándara, J. A. Reimer, O. M. Yaghi, J. Am. Chem. Soc., 2014, 136, 8863-8866.

- High Methane Storage Capacity in Aluminum Metal-Organic Frameworks, F. Gándara, H. Furukawa, S. Lee, O. M. Yaghi, J. Am. Chem. Soc., 2014, 136, 5271-5274.

- Water Adsorption in Porous Metal-Organic Frameworks and Related Materials, H. Furukawa, F. Gándara, Y.-B. Zhang, J. Jiang, W. L. Queen, M. R. Hudson, O. M. Yaghi, J. Am. Chem. Soc., 2014, 136, 4369-4381.

- Designed Amyloid Fibers as Materials for Selective Carbon Dioxide Capture, D. Li, H. Furukawa, H. Deng, C. Liu, O. M. Yaghi, D. S. Eisenberg, Proc. Natl. Acad. Sci. U.S.A., 2014, 111, 191-196.

- Storage of Hydrogen, Methane, and Carbon Dioxide in Highly Porous Covalent Organic Frameworks for Clean Energy Applications, H. Furukawa, O. M. Yaghi, J. Am. Chem. Soc., 2009, 131, 8875-8883.

- Highly Efficient Separation of Carbon Dioxide by a Metal-Organic Framework Replete with Open Metal Sites, D. Britt, H. Furukawa, B. Wang, T. G. Glover, O. M. Yaghi, Proc. Natl. Acad. Sci. U.S.A., 2009, 106, 20637-20640.

- Exceptional H2 Saturation Uptake in Microporous Metal-Organic Frameworks, A. G. Wong-Foy, A. J. Matzger, O. M. Yaghi, J. Am. Chem. Soc., 2006, 128, 3494-3495.

- Design and Synthesis of an Exceptionally Stable and Highly Porous Metal-Organic Framework, H. Li, M. Eddaoudi, M. O'Keeffe, O. M. Yaghi, Nature, 1999, 402, 276-279.

- Establishing Microporosity in Open Metal-Organic Frameworks: Gas Sorption Isotherms for Zn(BDC) (BDC=1,4-Benzenedicarboxylate), H. Li, M. Eddaoudi, T. L. Groy, O. M. Yaghi, J. Am. Chem. Soc., 1998, 120, 8571-8572.

MULTI-FUNCTIONAL MATERIALS
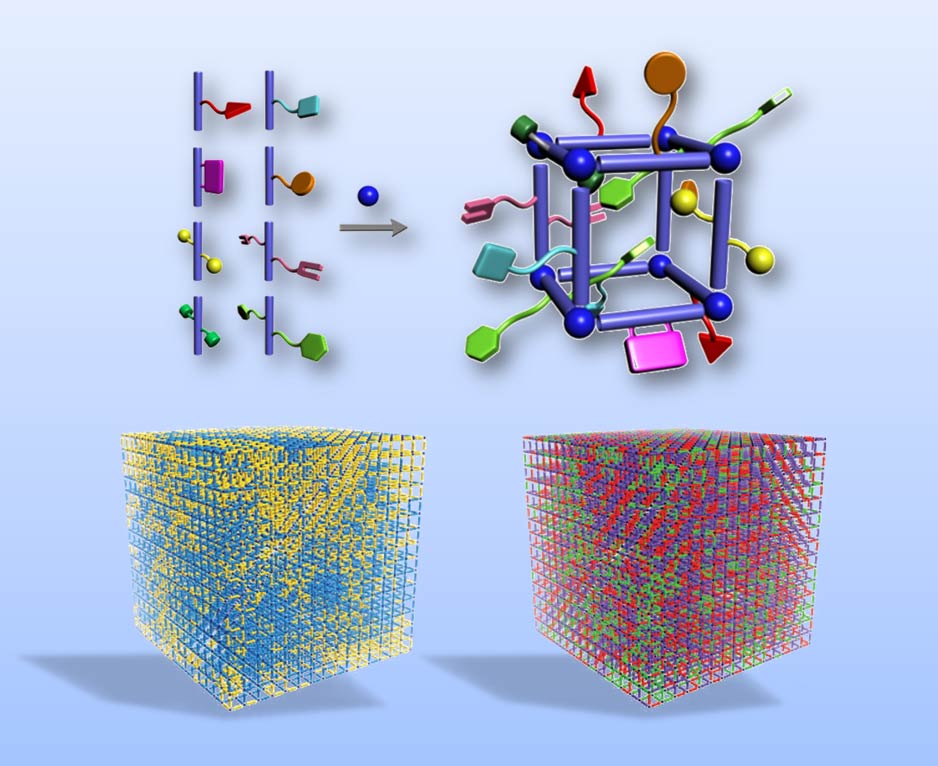
| The Yaghi group is working extensively on developing materials capable of counting and sorting molecules as well as coding for properties. The figure shows how our research group can achieve the assembly of multiple building units having multiple functionalities into a structure to make multivariate metal-organic frameworks (MTV-MOFs). These materials have pores decorated with sequences of functionalities, which we believe, code for specific properties. The bottom two structures show how such functionalities can be arranged in small intermingled clusters while other functionalities can be thoroughly mixed to give highly specific carbon capture properties, and by-product free catalysis. |
Beyond MOFs, COFs and ZIFs, and related porous crystals mentioned above we study ways of creating new materials based on the flexibility with which the building units can be varied. Among these are multivariate metal-organic frameworks (MTV-MOFs): 3D structures in which we introduce a large number of functionalities such that a MOF incorporates a multivariable arrangement of these functional groups and yields heterogeneity only found in biological systems. Since we can vary the type and ratio of functionalities, MTV-MOFs can be designed to have regions of a targeted functionality intermingled with regions of another and therefore have polar and non-polar regions juxtaposed as found in proteins. These systems raise interesting questions dealing with controlled complexity: what is the sequence of functionality within the MTV-MOFs and could these sequences be identified and designed to code for specific properties?
- MTV-MOFs: synthesis, X-ray structure analysis, spectroscopic characterization, and theoretical studies of their complexity and coding of information (Read more: UCLA Newsroom website)
- MTV-MOFs and the design of compartments which are linked but operate differently
- MTV-MOFs and their design as hosts to protein guests for biomedical imaging and drug delivery
- Covalent organic frameworks and incorporation of multi-variability and functional group sequences and codons in their backbone structure
- Covalent organic frameworks as modules for storage of gases to supply biological ensembles in the conversion of carbon dioxide to fuels
- Design, synthesis and study of ultrahigh porosity MTV-MOF materials in which heterogeneity imparts properties beyond those of the parts or their sum (whole operates better than the sum of the parts) (Read more: R&D Magazine)
- Synthesis and Characterization of Metal-Organic Framework-74 Containing 2, 4, 6, 8, and 10 Different Metals, L. J. Wang, H. Deng, H. Furukawa, F. Gándara, K. E. Cordova, D. Peri, O. M. Yaghi, Inorg. Chem., 2014, 53, 5881-5883.

- Mapping of Functional Groups in Metal-Organic Frameworks, X. Kong, H. Deng, F. Yan, J. Kim, J. A. Swisher, B. Smit, O. M. Yaghi, J. A. Reimer, Science, 2013, 341, 882-885.

- Multiple Functional Groups of Varying Ratios in Metal-Organic Frameworks, H. Deng, C. J. Doonan, H. Furukawa, R. B. Ferreira, J. Towne, C. B. Knobler, B. Wang, O. M. Yaghi, Science, 2010, 327, 846-850.

INORGANIC CHEMISTRY
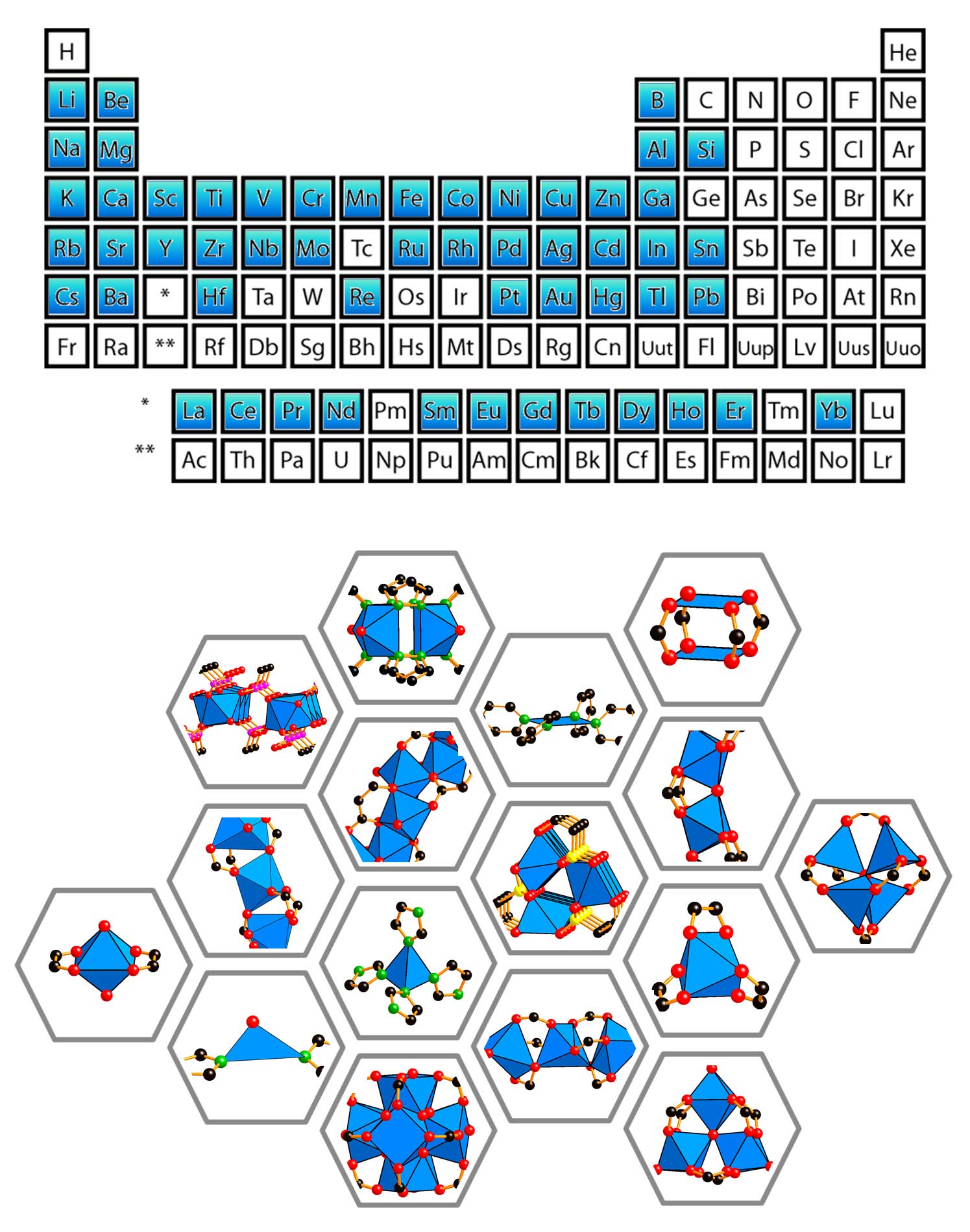
| The Yaghi group has taken metal-oxide clusters from the almost forgotten inorganic literature and succeeded in linking them with organic molecules to make porous metal-organic frameworks. Students in our group experience inorganic synthesis of clusters using elements from almost every corner of the periodic table, and their assembly using solution and solid-state techniques. We also functionalize the MOF structure with coordination and organometallic complexes for metal-based catalysis (e.g. activation of methane and water splitting for hydrogen generation). |
Our research is focused on using metal complexes and metal-oxide clusters as building units in the synthesis of MOFs. These inorganic units are copolymerized with organic linkers to make MOFs and related porous crystals. An important direction pertains to exposing metal coordination sites within the pores. Here, after the MOF structure formation, terminal ligands are found on the metal connectors. These ligands can be removed with full preservation of the structure, thereby leading to open metal sites in low coordinated metals. The electronic and steric nature of these open metal sites makes them ideal for enhanced gas storage and Lewis acid-based catalysis.
- Use of metal-oxide and nitride clusters as building units for robust frameworks with high thermal and chemical stability
- Design of open metal sites in the interior of MOFs for enhanced hydrogen storage, methane storage, selective carbon capture, and Lewis acid catalysis
- Covalent post-synthetic modification of MOFs and COFs with coordination and organometallic complexes for solar conversion and production of clean fuels
- Design of precise molecular architectures (enzyme-like) within the interior of MOFs for highly selective separations and reactions
- Synthesis and electronic properties of new forms of 3D carbon, boron, carbon nitride, boron nitride, and silicon carbide
- Synthesis, Structure, and Metalation of Two New Highly Porous Zirconium Metal-Organic Frameworks, W. Morris, B. Volosskiy, S. Demir, F. Gándara, P. L. McGrier, H. Furukawa, D. Cascio, J. F. Stoddart, O. M. Yaghi, Inorg. Chem., 2012, 51, 6443-6445.

- Metal Insertion in a Microporous Metal-Organic Framework Lined with 2,2'-Bipyridine, E. D. Bloch, D. Britt, C. J. Doonan, F. J. Uribe-Romo, H. Furukawa, J. R. Long, O. M. Yaghi, J. Am. Chem. Soc., 2010, 132, 14382-14384.

- Highly Efficient Separation of Carbon Dioxide by a Metal-Organic Framework Replete with Open Metal Sites, D. Britt, H. Furukawa, B. Wang, T. G. Glover, O. M. Yaghi, Proc. Natl. Acad. Sci. U.S.A., 2009, 106, 20637-20640.

- Isoreticular Metalation of Metal-Organic Frameworks, C. J. Doonan, W. Morris, H. Furukawa, O. M. Yaghi, J. Am. Chem. Soc., 2009, 131, 9492-9493.

- Exceptional Chemical and Thermal Stability of Zeolitic Imidazolate Frameworks, K. S. Park, Z. Ni, A. P. Côté, J. Y. Choi, R. D. Huang, F. J. Uribe-Romo, H. K. Chae, M. O'Keeffe, O. M. Yaghi, Proc. Natl. Acad. Sci. U.S.A., 2006, 103, 10186-10191.

- Strategies for Hydrogen Storage in Metal-Organic Frameworks, J. L. C. Rowsell, O. M. Yaghi, Angew. Chem. Int. Ed., 2005, 44, 4670-4679.

ORGANIC CHEMISTRY
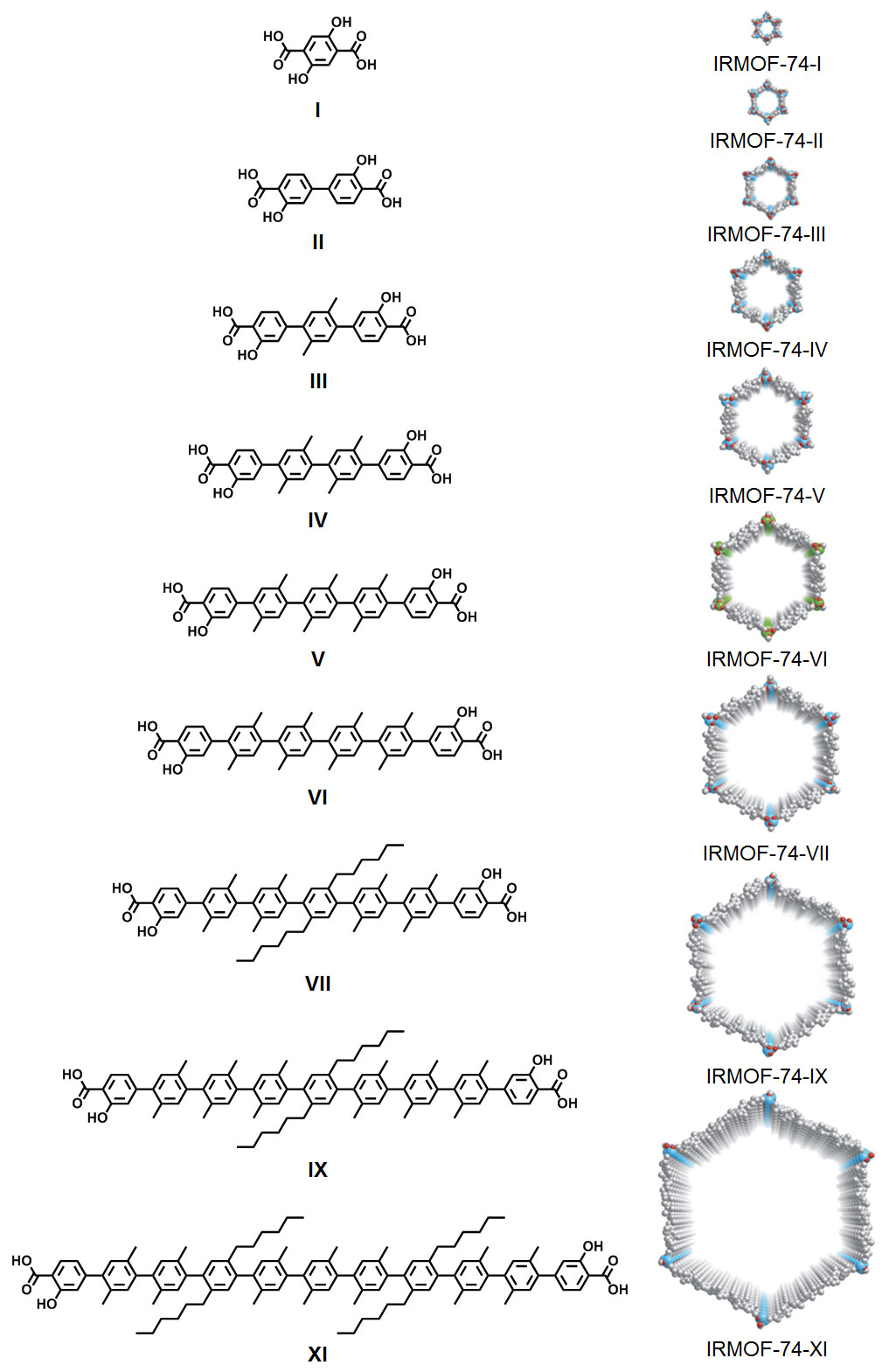
| Organic chemistry is central to the work being done in the Yaghi group. Typically, projects involve developing synthetic strategies for making organic links and functionalities capable of gas separation, carbon dioxide capture, methane storage, and catalysis. We also carry out extensive organic reaction chemistry in our efforts to post-synthetically modify the interior of the pores to produce precisely shaped architectures for highly specific binding of substrates. The example shown above is representative: progressively longer linkers were synthesized and made into MOFs having the largest pores ever made, enabling exceptional hydrogen storage, capture of carbon dioxide from flue gas, and inclusion of large molecules such as green fluorescent proteins, Vitamin B12, and hemoglobin. We are also pursuing the development of new forms of carbon, and new reactions for linking organic building units to make covalent organic frameworks. |
We work extensively on the synthesis of organic linkers for their use in metal-organic frameworks. These organic units are made from simple starting materials using efficient synthetic protocols. Their linking functionality is typical carboxyl groups but we are also interested in catecholes, beta-diketonates, imidazolates, triazolates and the like. Since these functionalities are charged, they form strong bonds to metal ions in the formation of extended porous networks. Another aspect of this work deals with the covalent organic chemistry we carry out on the frameworks after they form in order to functionalize the interior of the frameworks. Of course, all the reaction chemistry and the assembly of the covalent organic frameworks (COFs) involve intimate knowledge of organic reactions leading to C-B, C-N, C-O, and C-C bond formation.
- Design and synthesis of complex organic molecules and their use as "struts" in the assembly of MOFs and COFs
- Assembly of organic molecules into extended porous structures to make covalent organic frameworks
- Use of amino acids in building peptide functionalities within the interior of MOFs and COFs and their design for carbon capture
- Large pore openings for incorporation of drug molecule guests in MOF and COF hosts
- Crystals as molecules: post-synthetic covalent organic reactions for transforming the properties of MOFs, COFs, and ZIFs after crystal formation without losing their porosity
- Large-Pore Apertures in a Series of Metal-Organic Frameworks, H. Deng, S. Grunder, K. E. Cordova, C. Valente, H. Furukawa, M Hmadeh, F. Gándara, A. C. Whalley, Z. Liu, S. Asahina, H. Kazumori, M. O'Keeffe, O. Terasaki, J. F. Stoddart, O. M. Yaghi, Science, 2012, 336, 6084, 1018-1023.

- Metal-Organic Frameworks Incorporating Copper-Complexed Rotaxanes, A. Coskun, M. Hmadeh, G. Barin, F. Gándara, Q. Li, E. Choi, N. L. Strutt, D. B. Cordes, A. M. Z. Slawin, J. F. Stoddart, J.-P. Sauvage, O. M. Yaghi, Angew. Chem. Int. Ed., 2012, 51, 2160-2163.

- Covalent Organic Frameworks with High Charge Carrier Mobility, S. Wan, F. Gándara, A. Asano, H. Furukawa, A. Saeki, S. K. Dey, L. Liao, M. W. Ambrogio, Y. Y. Botros, X. Duan, S. Seki, J. F. Stoddart, O. M. Yaghi, Chem. Mater., 2011, 23, 4094-4097.

- A Metal-Organic Framework with Covalently Bound Organometallic Complexes, K. Oisaki, Q. Li, H. Furukawa, A. U. Czaja, O. M. Yaghi, J. Am. Chem. Soc., 2010, 132, 9262-9264.

- Docking in Metal-Organic Frameworks, Q. Li, W. Zhang, O. Š. Miljanić, C.-H. Sue, Y.-L. Zhao, L. Liu, C. B. Knobler, J. F. Stoddart, O. M. Yaghi, Science, 2009, 325, 855-859.

- Designed Synthesis of 3D Covalent Organic Frameworks, H. M. El-Kaderi, J. R. Hunt, J. L. Mendoza-Cortés, A. P. Côté, R. E. Taylor, M. O'Keeffe, O. M. Yaghi, Science, 2007, 316, 268-272.

- Porous, Crystalline, Covalent Organic Frameworks, A. P. Côté, A. I. Benin, N. W. Ockwig, A. J. Matzger, M. O'Keeffe, O. M. Yaghi, Science, 2005, 310, 1166-1170.

NANOSCIENCE
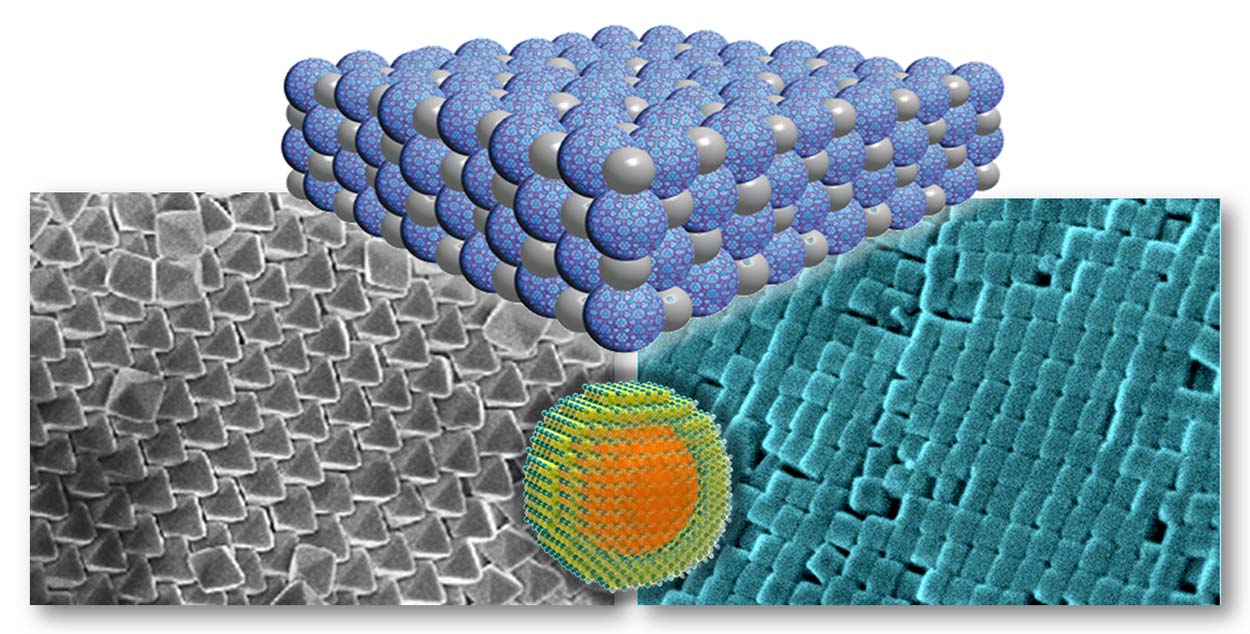 |
| The control in composition, structure and metrics of frameworks is being translated into the nano-regime. Our approach is to mix variously shaped nanocrystals into superlattices for light conversion to fuels, supercapacitors and biomedical applications. Students in this program learn how to navigate between diverse fields such as colloids, organic, inorganic and materials, and become well-versed in many related physical techniques such as scanning electron microscopy, high resolution tunneling electron microscopy, small angle x-ray scattering, and powder x-ray diffraction. |
We have recently succeeded in making nanocrystals of a large number of MOFs. The research in the group encompasses the synthesis of nanocrystals of various porous multivariate (MTV) MOFs in which the pores are decorated with a large number of organic functional groups. This research is expanding in several directions including: (a) control of composition, atomic structure, and metrics of the nanocrystals to design devices capable of hybrid properties such as electronic conductivity and solar energy conversion. (b) The fact that the pores of these MTV-MOF nanocrystals have a large variation of functionalities leads us to investigate whether the sequence of the functionalities are unique and thus could be used to code for specific properties. (c) Many of these nanocrystals are of the same size regime as proteins and other biological molecules, an aspect that is helping us in designing MTV-MOF nanocrystals for biomedical imaging and drug transport.
- Heterolites as new supercrystals for charge separation in light driven conversion involving reactions of methane, carbon dioxide and water to produce clean fuels
- NanoMOFs as supercapacitors with ultrahigh capacitance, charge mobility and lifetime including device design and measurement techniques
- Designed NanoMOF coatings of inorganic nanocrystals, nanowires and nanoparticles for observing single molecule events and conversion of methane and carbon dioxide to fuels
- Assembly of nanocrystalline MOFs on surfaces using layer-by-layer techniques for gated adsorption and separation membranes (Read more: C&EN)
- Metal Nanocrystals Embedded in Single Nanocrystals of MOFs Give Unusual Selectivity As Heterogeneous Catalysts, K. Na, K. M. Choi, O. M. Yaghi, G. A. Somorjai, NANO Lett., 2014, 14, 5979-5983.

- Supercapacitors of Nanocrystalline Metal-Organic Frameworks, K. M. Choi, H. M. Jeong, J. H. Park, Y.-B. Zhang, J. K. Kang, O. M. Yaghi, ACS Nano, 2014, 8, 7451-7457.

- Multiple Functional Groups of Varying Ratios in Metal-Organic Frameworks, H. Deng, C. J. Doonan, H. Furukawa, R. B. Ferreira, J. Towne, C. B. Knobler, B. Wang, O. M. Yaghi, Science, 2010, 327, 846-850.

- Crystal Structure, Dissolution, and Deposition of a 5 nm Functionalized Metal-Organic Great Rhombicuboctahedron, H. Furukawa, J. Kim, K. E. Plass, O. M. Yaghi, J. Am. Chem. Soc., 2006, 128, 8398-8399.

NANO-MOF SUPERCAPACITORS

| A transparent film of a MOF made into a supercapacitor device exhibits exceptional capacity and lifetime. The facility with which MOF materials can be designed allows the Yaghi group to survey the landscape of structure space in its effort to identify the best performing MOF. This also leads to identification of specific structure attributes, metrics and functionalities most favored for this application. |
Electrochemical capacitors or supercapacitors are an important class of energy storage devices because of their high power density. Porous carbon materials are commercial supercapacitors that operate by storing charge on electrochemical double layers. In contrast pseudocapacitors, typically made from metal oxides, store charge by redox reactions. Each of these classes of supercapacitor has advantages and disadvantages: carbon-based materials operate at very high charge/discharge rate with long lifecycle but have low capacitance, while metal oxide materials have high capacitance but their redox reactions lead to low lifecycle. Our group is working on ways of bridging the gap separating these two classes of supercapacitors using nano-MOFs. We examine different MOF compounds made in their nanocrystalline form. The measurements are done on thin film devices prepared from these nanocrystalline MOFs.
- Supercapacitor device construction using nano-MOFs and testing their ion-transport properties
- Examination of nano-MOFs with variable organic functionality, pore metrics and size of nanocrystals
- Design, synthesis and study of charge storage capacity in electronically conducting nano-MOFs
- Developing strategies for making nano-MOF superlattices and their use as membranes for separation of charged ions for application in water desalination
- Supercapacitors of Nanocrystalline Metal-Organic Frameworks, K. M. Choi, H. M. Jeong, J. H. Park, Y.-B. Zhang, J. K. Kang, O. M. Yaghi, ACS Nano, 2014, 8, 7451-7457.

- New Porous Crystals of Extended Metal-Catecholates, M. Hmadeh, Z. Lu, Z. Liu, F. Gándara, H. Furukawa, S. Wan, V. Augustyn, R. Chang, L. Liao, F. Zhou, E. Perre, V. Ozolins, K. Suenaga, X. Duan, B. Dunn, Y. Yamamto, O. Terasaki, O. M. Yaghi, Chem. Mater., 2012, 24, 3511-3513.

- Porous, Conductive Metal-Triazolates and Their Structural Elucidation by the Charge-Flipping Method, F. Gándara, F. J. Uribe-Romo, D. K. Britt, H. Furukawa, L. Lei, R. Cheng, X. Duan, M. O'Keeffe, O. M. Yaghi, Chem. Eur. J., 2012, 18, 10595-10601.

- Covalent Organic Frameworks with High Charge Carrier Mobility, S. Wan, F. Gándara, A. Asano, H. Furukawa, A. Saeki, S. K. Dey, L. Liao, M. W. Ambrogio, Y. Y. Botros, X. Duan, S. Seki, J. F. Stoddart, O. M. Yaghi, Chem. Mater., 2011, 23, 4094-4097.

BIOINSPIRED MATERIALS
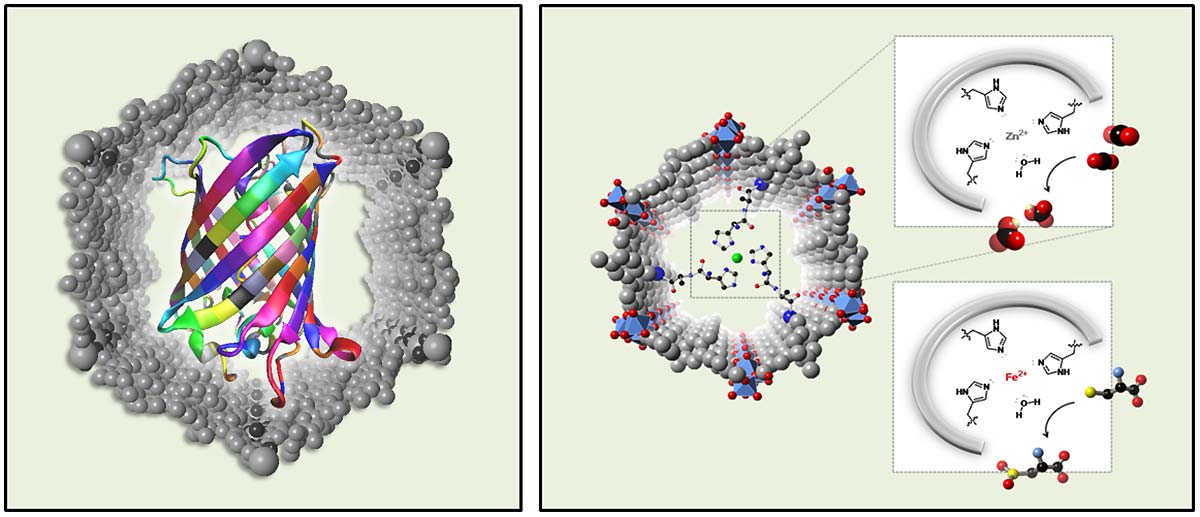 |
| The Yaghi group is working on several areas where concepts from biology are transferred into synthetic materials. On the left, MOFs are designed to be capable of trapping large proteins and ordering them in the pores to identify their structure and function. This applies also to crystallization of metabolites and peptides. On the right are functional groups (histidine) covalently arranged in the pore of a MOF to yield an intricate pocket for fixing carbon dioxide. |
MOF chemistry has matured to the point where the composition, structure, functionality, porosity, and metrics of a metal-organic structure can be designed for a specific application. This precise control over the assembly of materials is expected to propel this field further into new realms of synthetic chemistry in which far more sophisticated materials may be accessed. In our group we have begun to use concepts known in biology to make highly sophisticated materials. Research is focused on making and studying having (a) compartments linked together to operate separately, yet function synergistically; (b) dexterity to carry out parallel operations; (c) ability to count, sort, and code information; and (d) capability of dynamics with high fidelity.
- Materials beyond: a vast space of structures in which the backbone is made up from multiple different building units whose functionalities are arranged into specific sequences (Read more: Mother Nature Network)
- MOCAs (metal-organic complex arrays) are materials made of peptides decorated with arrays of metal ions which are arranged in very specific sequences to give sequence-dependent properties (Read more: C&EN)
- Design of complex artificial system capable of dynamics, yet remain robust. This is carried out using mechanical bonds as linkages in framework materials (Read more: C&EN, RSC website)
- Synthesis of metal-organic polymers capable of folding into various geometries to carry out reactions and dynamics
- Designed Amyloid Fibers as Materials for Selective Carbon Dioxide Capture, D. Li, H. Furukawa, H. Deng, C. Liu, O. M. Yaghi, D. S. Eisenberg, Proc. Natl. Acad. Sci. U.S.A., 2014, 111, 191-196.

- The Chemistry and Applications of Metal-Organic Frameworks, H. Furukawa, K. E. Cordova, M. O'Keeffe, O. M. Yaghi, Science, 2013, 341, 1230444.

- Isomers of Metal-Organic Complex Arrays, A. M. Fracaroli, K. Tashiro, O. M. Yaghi, Inorg. Chem., 2012, 51, 6437-6439.

- Synthesis of Metal-Organic Complex Arrays, P. Vairaprakash, H. Ueki, K. Tashiro, O. M. Yaghi, J. Am. Chem. Soc., 2011, 133, 759-761.

- Robust Dynamics, H. Deng, M. A. Olson, J. F. Stoddart, O. M. Yaghi, Nature Chem., 2010, 2, 439-443.

CATALYSIS
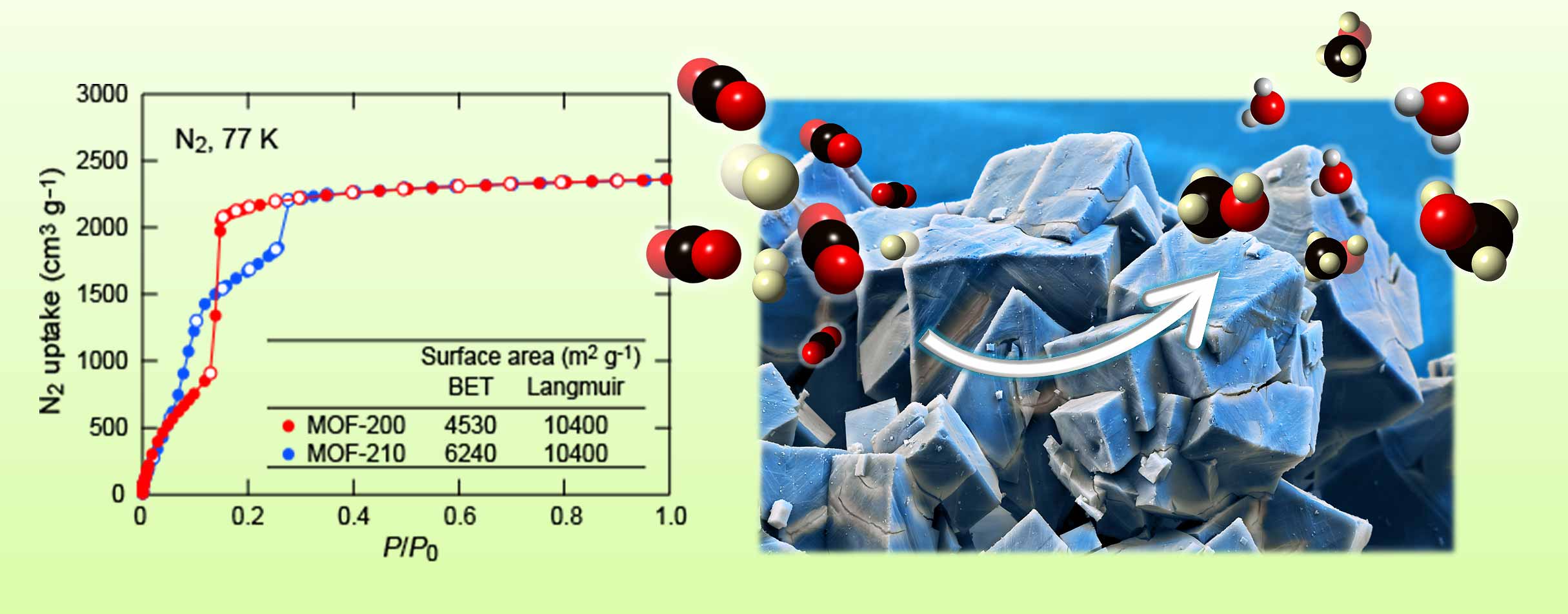 |
| A gas adsorption isotherm for some of the most reticular materials that indicate permanent porosity and ability to use the interior of MOFs for organic catalysis and positioning organometallic catalysts strategically such that reactions can be carried out with higher selectivity and efficiency. |
Our research group works closely with BASF-The Chemical Company on large-scale production of MOFs, COFs and ZIFs, and related materials. At present, MOFs have been commercialized by BASF under the label Basocubes and Basolites. Our studies in this direction take advantage of a long history of work on gas adsorption in MOFs, which originally was developed in our lab in 1998. We continue to develop MOFs and study their properties and applications in a wide range of fields as mentioned above but more recently we found they are ideally suited as catalysts for organic transformations..
- Catalysis within the pores of MOFs: use of Lewis acid sites and superacid sites in hydrolysis of organic substrates and isomerization of hydrocarbons
- Catalysis within active domains of MOFs: use of predesigned pockets within MOFs and ZIFs to convert methane and carbon dioxide to clean fuels
- Cyclization organic reactions carried out with high selectivity in MOF and COF frameworks
- Use of vanadium MOFs with rod-shaped building units to convert methane to acetic acid and other feedstock molecules
- Growth of metal nanoparticles within the pores of MOFs for carbon-carbon breaking reactions
- Metal Nanocrystals Embedded in Single Nanocrystals of MOFs Give Unusual Selectivity As Heterogeneous Catalysts, K. Na, K. M. Choi, O. M. Yaghi, G. A. Somorjai, NANO Lett., 2014, 14, 5979-5983.

- Superacidity in Sulfated Metal-Organic Framework-808, J. Jiang, F. Gándara, Y.-B. Zhang, K. Na, O. M. Yaghi, W. G. Klemperer, J. Am. Chem. Soc., 2014, 136, 12844-12847.

- A Multiunit Catalyst with Synergistic Stability and Reactivity: A Polyoxometalate-Metal Organic Framework for Aerobic Decontamination, J. Song, Z. Luo, D. K. Britt, H. Furukawa, O. M. Yaghi, K. I. Hardcastle, C. L. Hill, J. Am. Chem. Soc., 2011, 133, 16839-16846.

- Metal-Organic Frameworks of Vanadium as Catalysts for Conversion of Methane to Acetic Acid, A. Phan, A. U. Czaja, F. Gándara, C. B. Knobler, O. M. Yaghi, Inorg. Chem., 2011, 50, 7388-7390.

- Asymmetric Catalytic Reactions by NbO-Type Chiral Metal-Organic Frameworks, K. S. Jeong, Y. B. Go, S. M. Shin, S. J. Lee, J. Kim, O. M. Yaghi, N. Jeong, Chem. Sci., 2011, 2, 877-882.

HIGH-THROUGHPUT SYNTHESIS AND MATERIAINFORMATICS
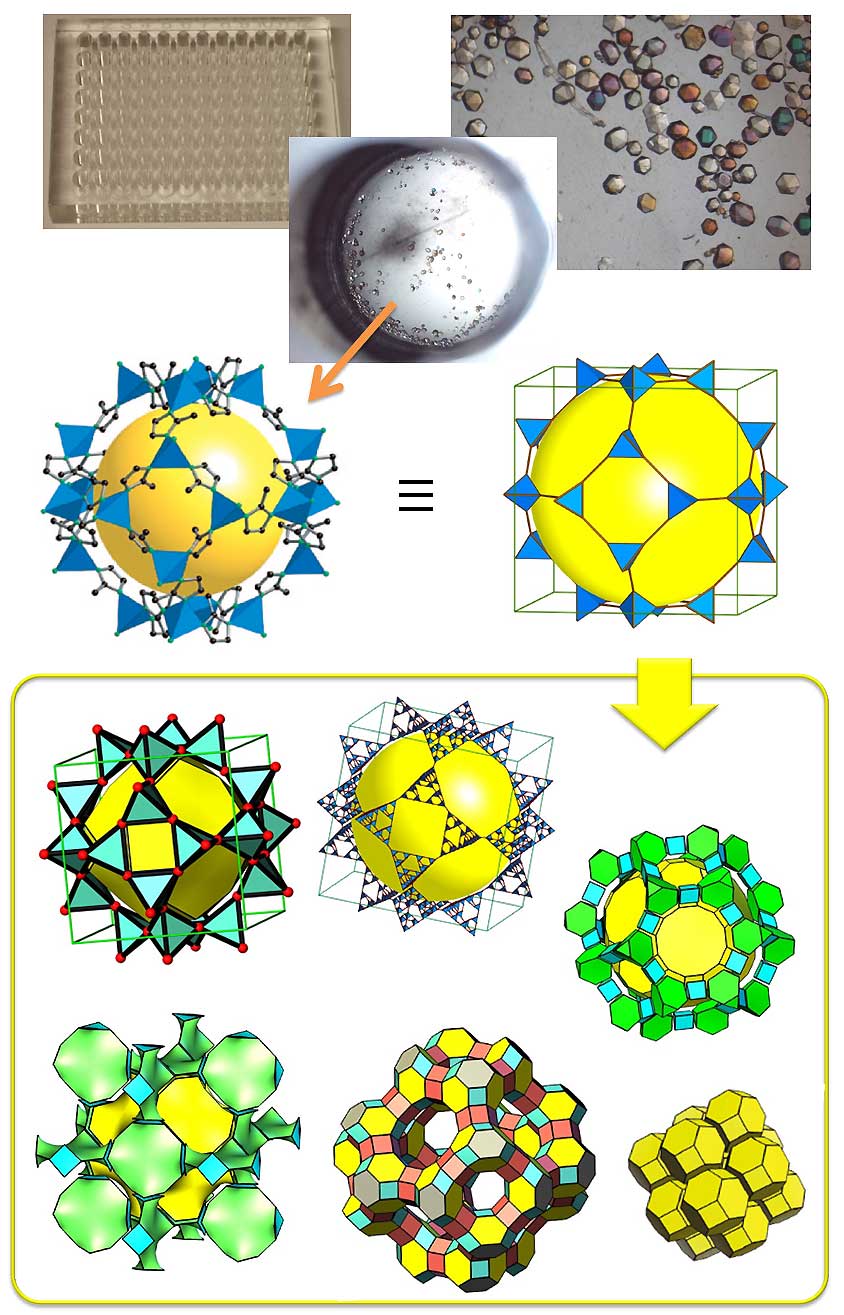
| The Yaghi lab is equipped with high-throughput equipment, which is used to discover new materials and screen for various properties as mentioned above. In the example shown here, over 30 new zeolitic imidazolate frameworks were discovered using this technique from only 9,600 reactions. |
During the past decade, interest has grown tremendously in the design and synthesis of crystalline materials constructed from molecular clusters linked by extended groups of atoms. Most notable are MOFs, in which polytopic organic linkers join polyatomic inorganic metal-containing clusters. The realization that MOFs could be designed and synthesized in a rational way from molecular building blocks led to the emergence of a discipline that we call reticular chemistry. Because of the explosive growth in this area, a need has arisen for a universal system of nomenclature, classification, identification, and retrieval of these topological structures. We are developing a system of symbols for the identification of three periodic nets of interest, and this system is now in wide use. In reticular chemistry, we now can, for a given set of building units, predict with some certainty, the structures which might result from the assembly of those building blocks. We then use high-throughput synthesis to survey the possible structure space and 'zoom-in' on the desirable phase and use for a particular application as explained above.
- Develop a system of nomenclature for the classification and identification of MOFs, COFs, and ZIFs
- Computation of topological possibilities for the assembly of building unit geometries and prediction of the powder x-ray diffraction pattern of the corresponding structures
- Survey of known MOFs and classification of their structure and gas adsorptive properties
- Use of high-throughput methods to discover new materials and to survey their properties including the development of pre-defined protocols for identifying good performing materials in gas adsorption, supercapacitors and catalysis (Read more: C&EN)
- Topological Analysis of Metal-Organic Frameworks with Polytopic Linkers and/or Multiple Building Units and the Minimal Transitivity Principle, M. Li, D. Li, M. O'Keeffe, O. M. Yaghi, Chem. Rev., 2014, 114, 1343-1370.

- Porous, Conductive Metal-Triazolates and Their Structural Elucidation by the Charge-Flipping Method, F. Gándara, F. J. Uribe-Romo, D. K. Britt, H. Furukawa, L. Lei, R. Cheng, X. Duan, M. O'Keeffe, O. M. Yaghi, Chem. Eur. J., 2012, 18, 10595-10601.

- Deconstructing the Crystal Structures of Metal-Organic Frameworks and Related Materials into Their Underlying Nets, M. O'Keeffe, O. M. Yaghi, Chem. Rev., 2012, 112, 675-702.

- High-Throughput Synthesis of Zeolitic Imidazolate Frameworks and Application to CO2 Capture, R. Banerjee, A. Phan, B. Wang, C. Knobler, H. Furukawa, M. O'Keeffe, O. M. Yaghi, Science, 2008, 319, 939-943.

- The Reticular Chemistry Structure Resource (RCSR) Database of, and Symbols for, Crystal Nets, M. O'keeffe, M. A. Peskov, S. J. Ramsden, O. M. Yaghi, Acc. Chem. Res., 2008, 41, 1782-1789.

- Reticular Chemistry: Occurrence and Taxonomy of Nets, and Grammar for the Design of Frameworks, N. W. Ockwig, O. Delgado-Friedrichs, M. O'Keeffe, O. M. Yaghi, Acc. Chem. Res., 2005, 38, 176-182.

- Gas Adsorption Sites in a Large-Pore Metal-Organic Framework, J. L. C. Rowsell, E. C. Spenser, J. Eckert, J. A. K. Howard, O. M. Yaghi, Science, 2005, 309, 1350-1354.